Pavilion Publishing and Media Ltd
Blue Sky Offices Shoreham, 25 Cecil Pashley Way, Shoreham-by-Sea, West Sussex, BN43 5FF, UNITED KINGDOM
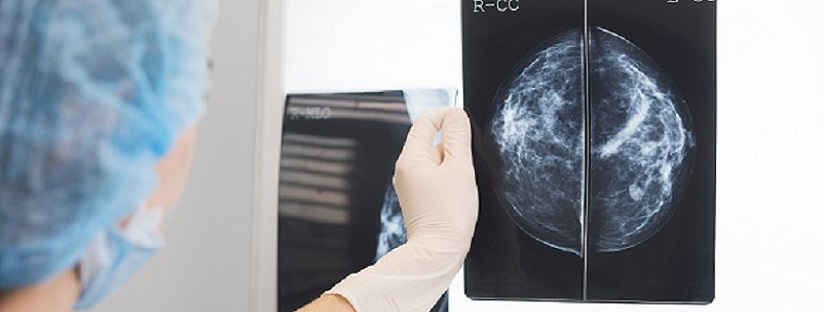
Breast cancer management in primary care
Although most women are aware that a lump can be a sign of breast cancer, awareness of other potential signs and symptoms among the public are poor. This article discusses risk factors and presentation of breast cancer in primary care and treatment of early disease.
---------------------------
This content is restricted to members of Pavilion Health Today. If you are an existing user, please log in. New users may register for free below.